Authors: Rizzo R., Russo D., Kurokawa, K., Sahu P., Lombardi, B., Supino D., Zhukovsky M.A., Vocat A., Pothukuchi P., Kunnathully V., Capolupo L., Boncompain G., Vitagliano C., Zito Marino F., Aquino G., Montariello D., Henklein P., Mandrich L., Botti G., Clausen H., Mandel U., Yamaji T., Hanada K., Budillon A., Perez F., Parashuraman S., Hannun Y.A.
EMBO Journal 2021
Keywords: cisternal maturation; Golgi; GOLPH3;
DOI: https://doi.org/10.15252/embj.2020107238
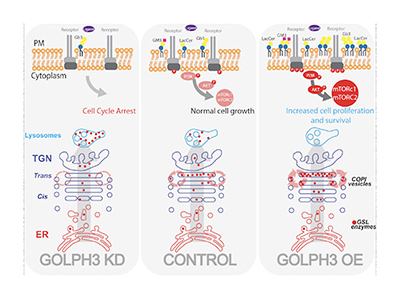
Abstract: Glycosphingolipids are important components of the plasma membrane where they modulate the activities of membrane proteins including signalling receptors. Glycosphingolipid synthesis relies on competing reactions catalysed by Golgi-resident enzymes during the passage of substrates through the Golgi cisternae. The glycosphingolipid metabolic output is determined by the position and levels of the enzymes within the Golgi stack, but the mechanisms that coordinate the intra-Golgi localisation of the enzymes are poorly understood. Here, we show that a group of sequentially-acting enzymes operating at the branchpoint among glycosphingolipid synthetic pathways binds the Golgi-localised oncoprotein GOLPH3. GOLPH3 sorts these enzymes into vesicles for intra-Golgi retro-transport, acting as a component of the cisternal maturation mechanism. Through these effects, GOLPH3 controls the sub-Golgi localisation and the lysosomal degradation rate of specific enzymes. Increased GOLPH3 levels, as those observed in tumours, alter glycosphingolipid synthesis and plasma membrane composition thereby promoting mitogenic signalling and cell proliferation. These data have medical implications as they outline a novel oncogenic mechanism of action for GOLPH3 based on glycosphingolipid metabolism.